OncoBeta GmbH are a privately held medical device company, specializing in the development and commercialization of state-of-the-art, innovative therapies utilizing epidermal radioisotope applications. Following their success in CV’s Corporate Excellence Awards 2019, we profiled the firm and caught up with Shannon Brown who provided us with a detailed glimpse into the innerworkings of the award-winning company.
Since their inception, OncoBeta GmbH have concentrated on an epidermal radioisotope therapy called the Rhenium-SCT® (Skin Cancer Therapy), targeting non-melanoma skin cancers. OncoBeta GmbH’s primary goal is to offer an effective, customized and non-invasive treatment, independent of tumor size, severity and localization. In addition to this, the company also aims to provide a fast, painless, single treatment, with no side effects and aesthetic results.
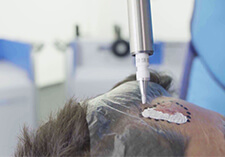
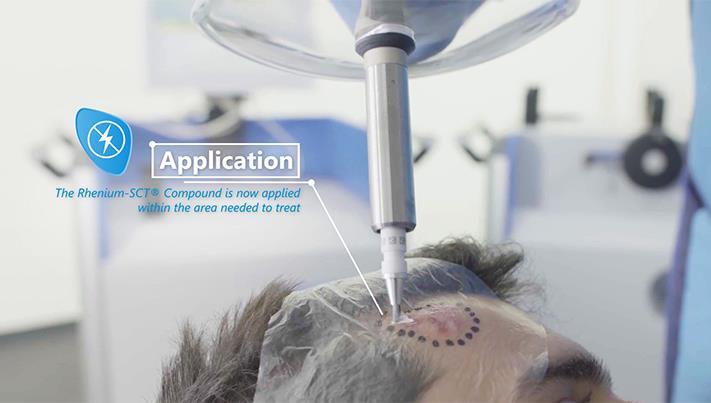
Rhenium-SCT® is a specialized state-of-the-art therapy utilizing the radioisotope Rhenium-188, a beta-emitter which in this case is used for the treatment of basal and squamous cell carcinomas of the skin. Going into further detail, Shannon begins by informing us more about Rhenium-SCT® and the medical working principle of using this particular therapy.
“The global incidence of non-melanoma skin cancers has been drastically increasing over the past few decades. Depending on the source, it is estimated that there are over five million non-melanoma skin cancer cases reported globally each year.”
“The medical working principle of the Rhenium-SCT® or epidermal radioisotope therapy is based on the local direct cell-killing effect of the beta-radiation, which triggers both the localized destruction of cancer cells and local reactions of the immune system of the body to repair itself.”
Today, OncoBeta GmbH is helping to redefine patient experiences, patient outcomes, patient expectations, while redefining the approach to treating non-melanocytic skin cancer (NMSC), one of the most common cancers. Patients who suffer from NMSCs are often left with standard care options, which are either painful, non-aesthetically desired, or are invasive procedures in nature. OncoBeta GmbH is part of the story trying to rectify this, as Shannon explains.
“With our therapeutic for NMSC – the Rhenium-SCT®, OncoBeta GmbH has a scalable, highly effective, clinically proven and desirable solution for NMSC patients, that improves overall patient outcomes and experiences. Scientifically, OncoBeta GmbH’s Rhenium-SCT® harnesses the radioisotope Rhenium-188 for clinical purposes in a safe, stable, and repeatable manner. Our manufacturing process, logistics network, and innovative approach to matching patient needs with our supply base are well defined. Overall, we are ensuring scalability and patient access to our therapeutic solution, ensuring patients a better future.”
Working behind the scenes to deliver these award-winning therapy treatments, are the dedicated, passionate and hard-working team which forms the backbone of OncoBeta GmbH.
When discussing the internal culture, Shannon is keen to highlight the significant role the team plays in the overall success of the company.
“Our highly dedicated team plays a large role in our success. The team works relentlessly to improve our treatment and to increase our current therapeutic portfolio, while constantly striving to develop additional non-invasive therapies in the areas of
dermatology and oncology for patients in need.”
Looking ahead to what the future holds for the company, the team at OncoBeta GmbH will continue to deliver their exceptional services, especially following their recent success in Corporate Vision’s Corporate Excellence Awards 2019 where they were righteously awarded the accolade Most Compassionate Skin Cancer Therapy Provider 2019. Bringing the interview to a close, Shannon signs off by revealing the future plans for the Rhenium-SCT®, as well as the industry challenges the team at OncoBeta GmbH are currently facing and how they hope to overcome them.
“The Rhenium-SCT® is presently available in Germany, Italy, Australia and South Africa. We anticipate expansionary activities in other European/ Non-European countries such as: France, United Kingdom, Spain, Austria, Switzerland, Scandinavian Countries, Canada, United States, SE Asia and Brazil scheduled over the next 2-3 years.
“OncoBeta GmbH is fighting from inside the healthcare apparatus to offer a better alternative for those suffering with NMSCs. This means being highly active in the NMSC therapeutic community, exchanging in dialogues to change the perspectives of clinicians, exchanging in dialogues to change clinicians minds about standard care and exchanging in dialogue to open up referral chains to other therapeutic alternatives, such as the Rhenium-SCT®. This is the fight of all innovations in the marketplace, to challenge the status quo. This is our biggest challenge, but one worth pursuing with the patient’s perspective always in mind.”
Company: OncoBeta GmbH
Web Address: www.oncobeta.com